Research
Our research programs are driven by questions relevant to human disease, spanning fundamental immunology through preclinical translation.
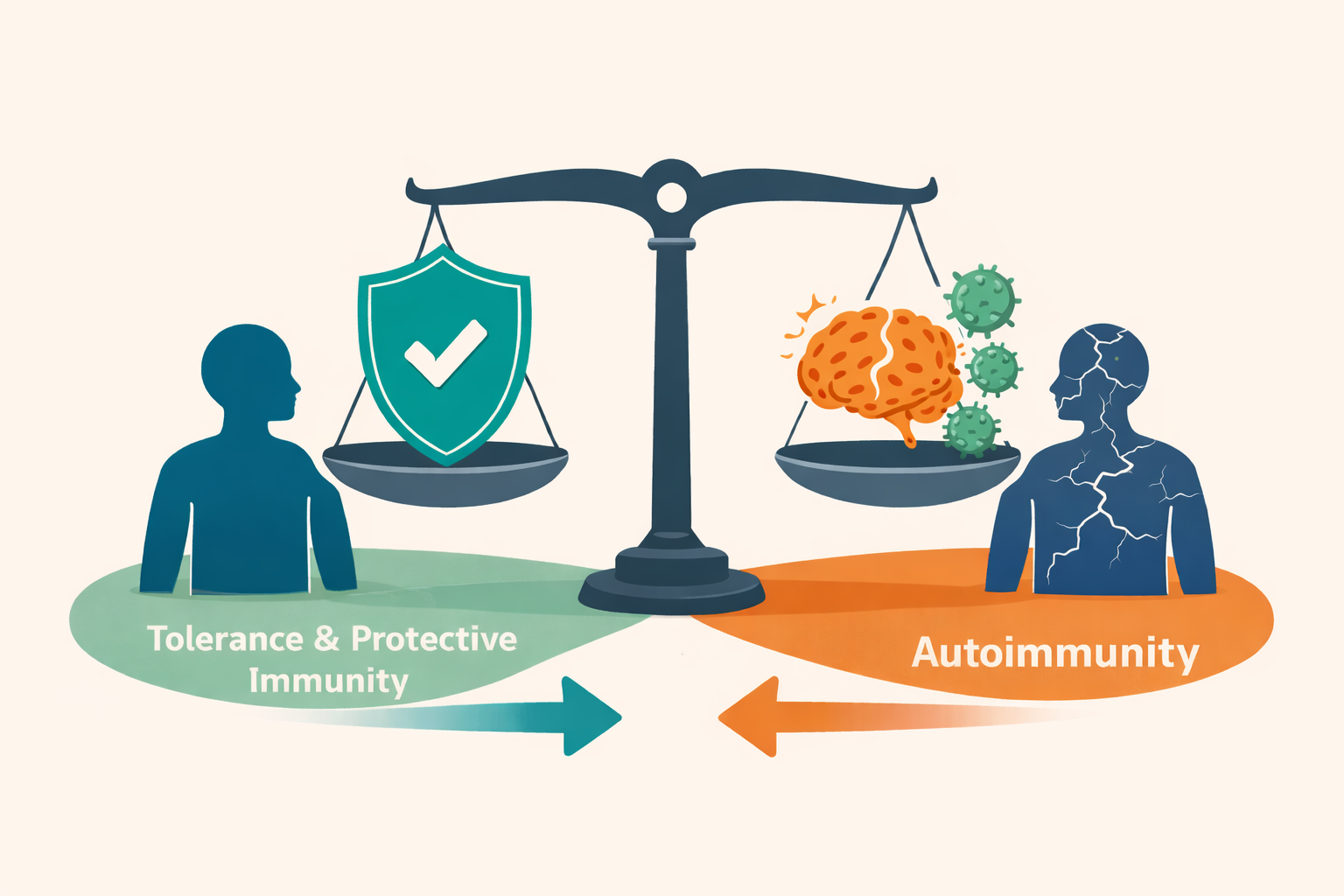
Immune Tolerance & Inflammatory Disease
The immune system must distinguish self from non-self, pathogen from commensal, and threat from noise, and it must do so across a lifetime of changing exposures. Our lab investigates how antigen-specific T cell responses are shaped during early immune education and how that homeostasis breaks down in autoimmune and inflammatory disease. We study synovial T cells in inflammatory arthritis, immune-related adverse events triggered by cancer immunotherapy, and the ways infectious exposures (including EBV, and potential triggers of Long COVID and ME/CFS) may initiate or amplify pathology through molecular mimicry or chronic antigen stimulation. Our goal is to understand not just that tolerance fails, but precisely where, when, and against what antigens, because that precision is what makes antigen-specific therapeutic intervention possible.
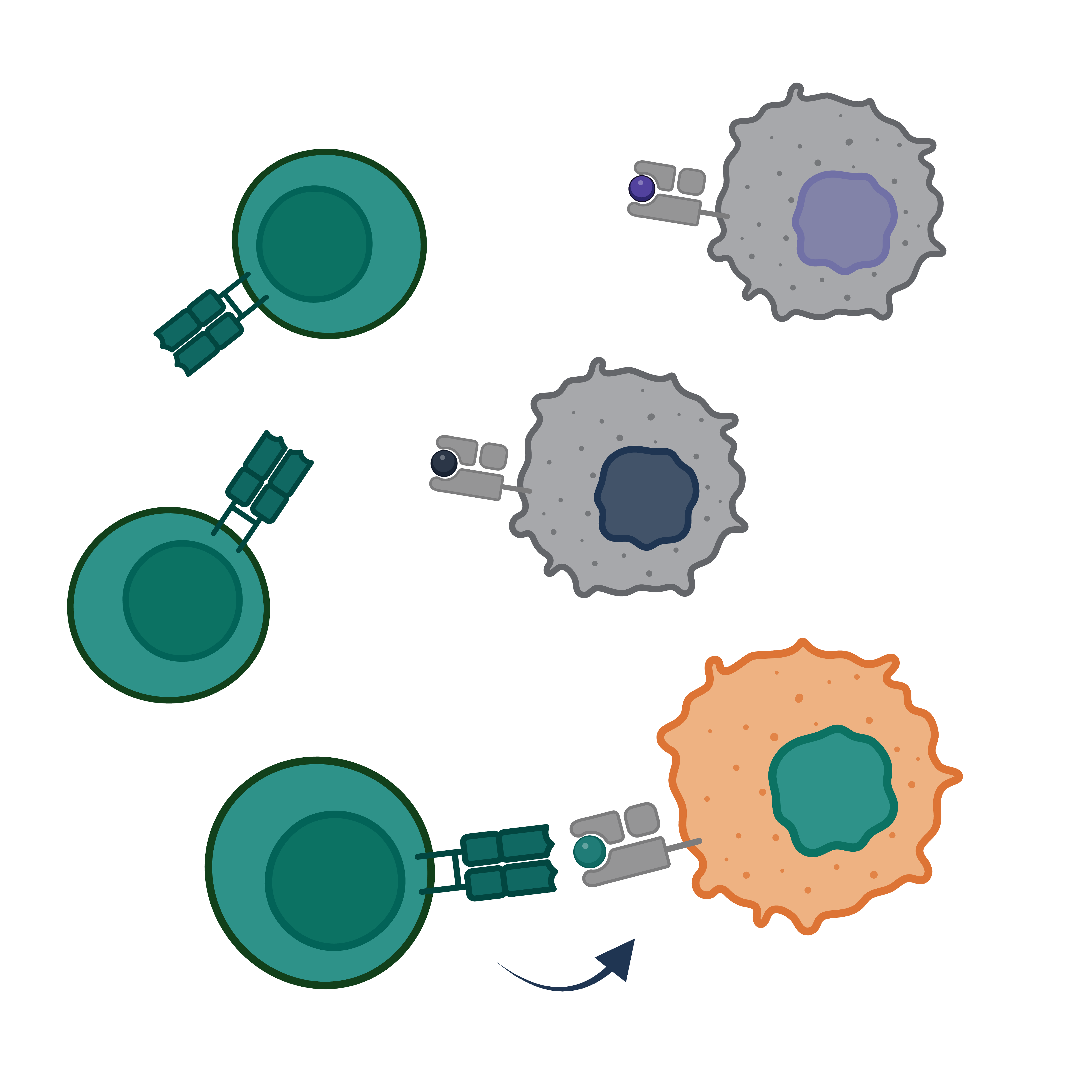
Antigen & Ligand Discovery
A T cell receptor (TCR) is only meaningful if you know what it sees. Our lab develops and applies experimental approaches to identify the specific antigens and ligands recognized by disease-associated T cells: the self-peptides driving autoimmunity, the microbial epitopes triggering cross-reactive responses, and the tumor-derived molecules that mark cancer cells as targets. We combine ribosome profiling, MHC immunopeptidomics, genome-scale antigen discovery methods like TCR-MAP, and pooled CRISPR screens targeting antigen processing and presentation. Together, these tools allow us to move systematically from a T cell phenotype or receptor sequence to the molecule it recognizes and the cellular pathway that generates it. Knowing the precise antigens recognized by T cells in disease is not only foundational to understanding immune pathology, but it also creates direct opportunities for therapeutic design. Defined antigens can anchor tolerizing vaccines aimed at quieting harmful autoimmune responses, protective vaccines can prime T cells against pathogens or tumors, and targeted cell therapies can exploit those specificities with precision. Defining these targets is foundational to everything else the lab does.
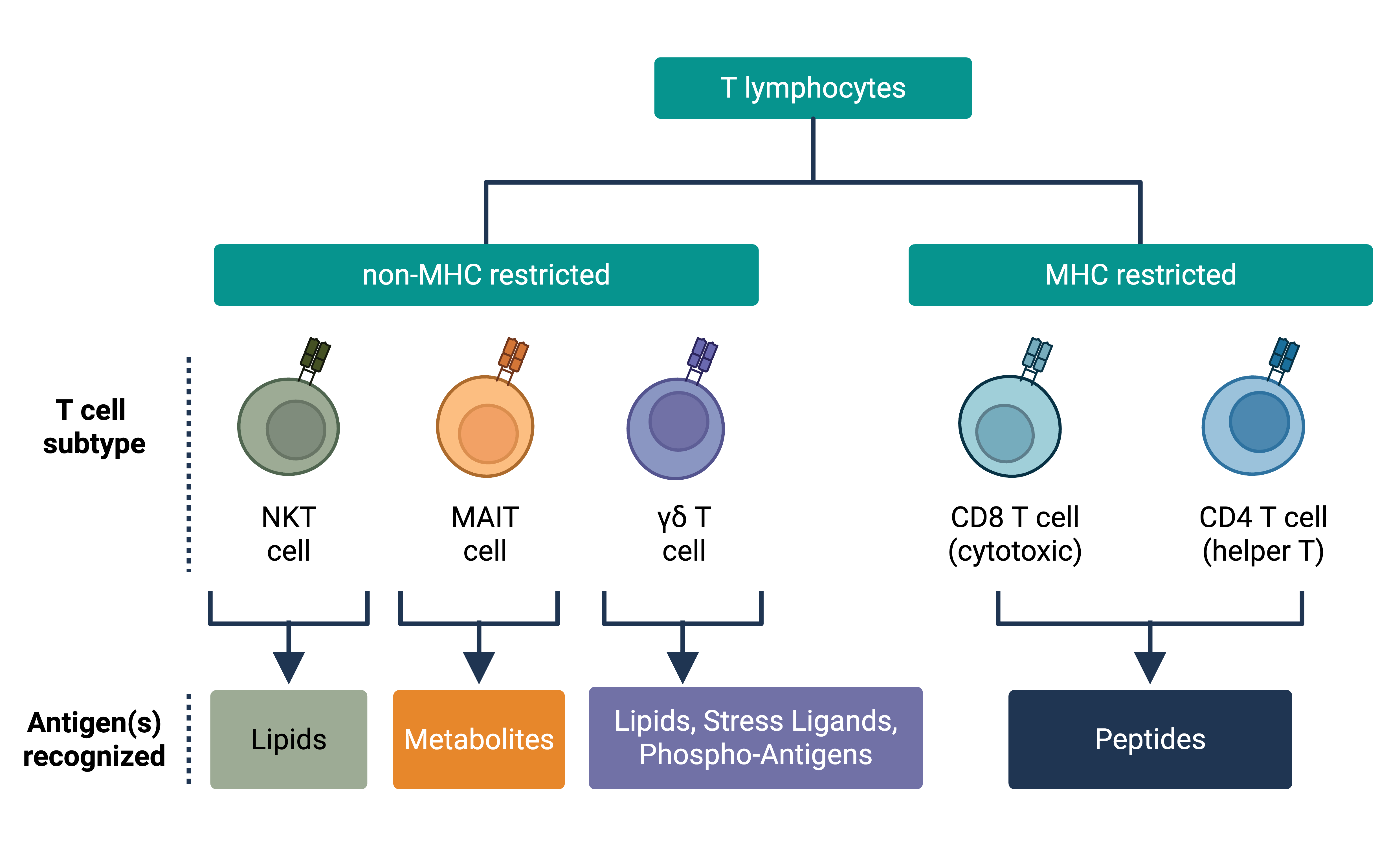
T Cell Specificity Technologies
Most tools for studying T cell recognition were built with classical αβ T cells in mind, leaving the specificity landscapes of γδ T cells, natural killer T (NKT) cells, and mucosal-associated invariant T (MAIT) cells comparatively unmapped. Our lab builds and adapts high-throughput platforms to query T cell specificity at scale and across the full diversity of T cell classes. This includes developing new screening systems, synthetic biology tools, and functional genomic approaches that can be applied broadly, not just to answer a single biological question, but to increase the resolution and throughput with which the field can interrogate immune recognition. In addition, our lab is developing new approaches to interrogate post-translationally modified (PTM) antigen landscapes, including citrullinated epitopes that are difficult to query at scale with conventional methods and that are relevant to autoimmune diseases such as rheumatoid arthritis. We see this technology development as way to improve knowledge, where better tools expand what questions become answerable, across autoimmunity, infection, and cancer.
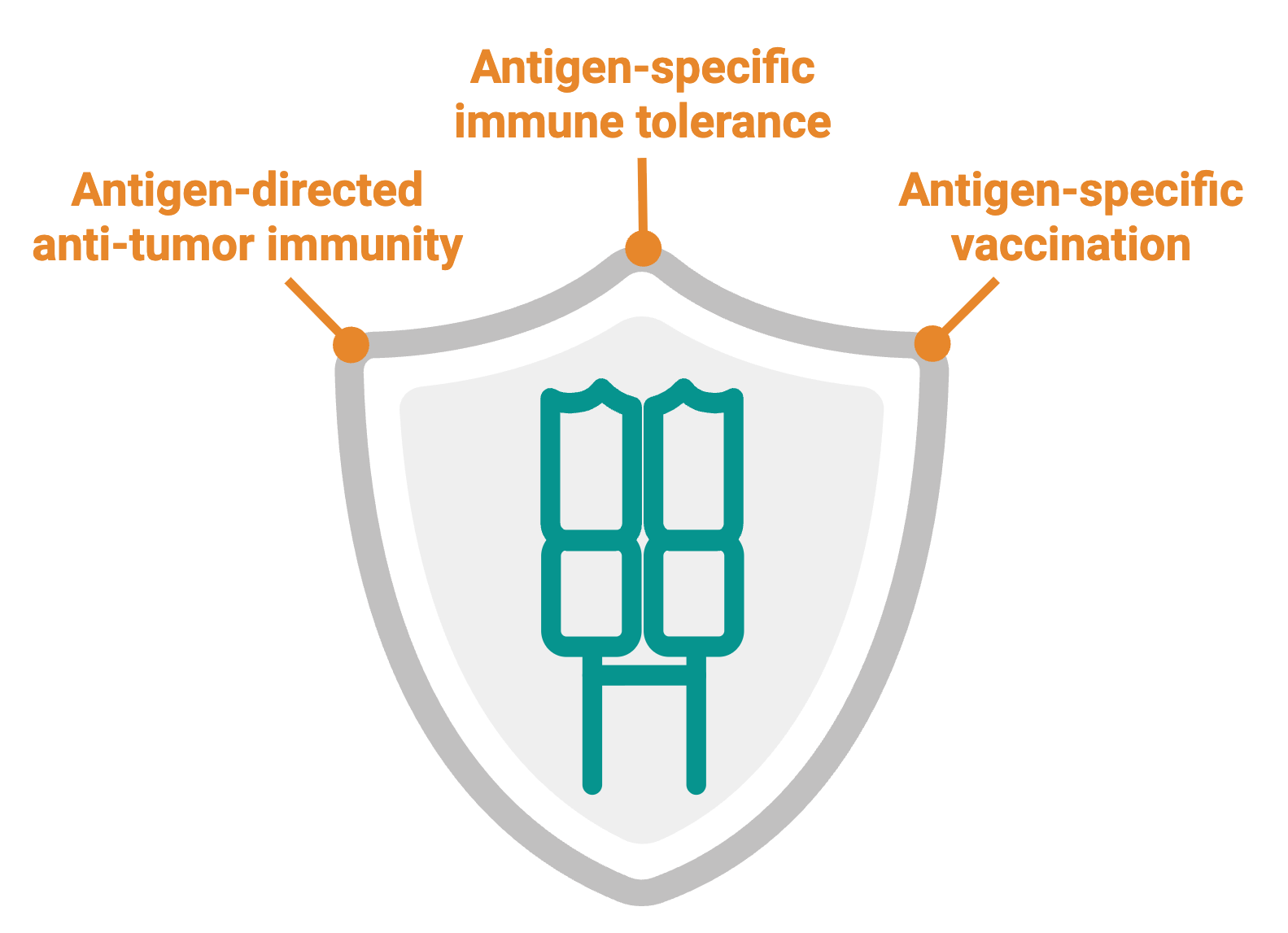
TCR Therapeutics & Precision Immunology
T cell receptors (TCRs) are not only readouts of immune history. They are molecular tools with therapeutic potential, and the antigens they recognize are blueprints for a new generation of targeted immune interventions. Our lab identifies TCRs with clinically relevant specificities, characterizes their recognition properties, and explores how they can inform or directly enable therapeutic development. In cancer, we focus on tumor-reactive receptors, including γδ TCRs, that could underpin next-generation cell therapies. In infectious disease, defining the T cell epitopes that correlate with protective immunity informs rational vaccine design and helps explain why some responses succeed where others fail. In autoimmunity, understanding which antigens drive pathogenic T cell responses, and which specificities are carried by protective regulatory T cells, opens the door to tolerizing vaccines and antigen-specific strategies that restore immune balance without broad immunosuppression. Across these settings, our work is grounded in human disease and patient-derived immune responses, and is motivated by the idea that immune specificity, once defined, can be turned into a therapeutic advantage.